Common Illnesses
Common Illness and Treatments: FIP UTI COCCIDIA
FIP
Feline Infectious Peritonitis is one of the most difficult challenges a breeder will face when breeding cats. Here are some resources to reference when researching the disease. These articles present a neutral and informative viewpoint and give the latest in FIP research.
Cornell University
Winn Feline Foundation
New “SNAP” test for FECV! (Only available for import to your vet!)
Potential New Treatment for Feline Infectious Peritonitis (FIP)
Of all of the infectious disease that cats can acquire, feline infectious peritonitis (FIP) is perhaps the most devastating. FIP is a viral disease caused by a type of virus called a coronavirus. Most cats are exposed to, and become infected with, the coronavirus as kittens.
At worst, kittens may get mild diarrhea. Many show no clinical signs at all. The immune system makes antibodies against the virus but does not eliminate it, and the virus continues to reside in the intestinal tract usually causing no further problems for the cat.
Occasionally, for reasons not fully understood, the harmless intestinal coronavirus can change or mutate, gaining the ability to leave the intestinal tract and cause horrible problems. The immune system tries to defeat the virus, but the virus manages to evade it. This mutated intestinal coronavirus is now the evil FIP-inducing coronavirus.
The FIP virus causes damage to blood vessels, allowing fluid to leak through the vessel walls. Effusions (collections of fluid) can develop in the abdominal cavity, chest cavity, and pericardium (the sac around the heart) resulting in what is referred to as the “wet” form of FIP. Some cats develop nodular accumulations of inflammatory cells called granulomas throughout many of the body’s organs. This form of the disease is known as the “dry” form of FIP.
Diagnosis of the disease is difficult because clinical signs are vague and non-specific. Most cats are young (usually less than one year), and show lethargy, weight loss, poor appetite, and a fever that doesn’t respond to antibiotics. A serum chemistry panel often only shows elevated protein (mainly in the form of increased globulins) unless the virus has begun to affect the kidneys or liver, in which case the liver and kidney parameters may be abnormal. The wet form is easier to diagnose because the presence of fluid in the abdomen or chest is relatively easy to detect and fluid analysis can give additional information supporting the diagnosis. The dry form remains one of the more challenging diseases for veterinarians to diagnose.
There is no simple blood test for FIP. Many laboratories offer veterinarians an “FIP test”, but these tests only measure antibodies to coronaviruses in general. It cannot distinguish whether the antibodies are there due to the cat being infected with the harmless intestinal version of the coronavirus, or the deadly FIP version of the coronavirus. A positive test does not mean that the cat has FIP. Until a rapid reliable test is developed that allows veterinarians to make a diagnosis, biopsy of the affected organs or tissues remains the only way to definitively diagnose FIP, although Auburn University offers an assay that they claim is very good for diagnosing FIP.
A commercial FIP vaccine is available, however, the efficacy of the vaccine remains questionable. The American Association of Feline Practitioners and the Academy of Feline Medicine, in their regularly published guidelines for feline vaccination, currently do not recommend the FIP vaccine at this time.
FIP is progressive and fatal. Cats with FIP tend to succumb to the disease rather quickly, in a few days or weeks. The “wet” form of FIP tends to progress faster than “dry” FIP. Treatment is generally symptomatic and supportive. Nutritional support, antibiotics, and corticosteroids may produce a temporary alleviation of clinical signs, but the disease invariably progresses.
A few years ago an article appeared in the Journal of Feline Medicine and Surgery about a potential new treatment for FIP. This was very exciting for us feline practitioners, because FIP is a truly devastating disease that leaves us totally helpless. In the study, 12 cats with FIP were given prednisolone and recombinant feline interferon. Four of the cats died relatively quickly. Four cats survived for a short period of time – between 2 and 5 months. The last four, however, showed prolonged survival. This was an uncontrolled study, but it was really all that we had.
Not long after the article came out, I diagnosed a cat with FIP. I mentioned the new study to the client, and they were interested. A problem: the interferon that we use in veterinary practice is recombinant human interferon. Recombinant feline interferon is not available in the United States. It’s available in the European Union. Well… I managed to score some; we won’t go into the details. I used it on the cat, but to no avail. The cat (a kitten actually) succumbed to the disease. Not long after that, a controlled study of the use of recombinant feline interferon in over 50 cats with FIP was published. The study concluded that recombinant feline interferon was ineffective in treating FIP. Darn it.
An article was recently published in the November 2009 Journal of Feline Medicine and Surgery on the use of polyprenyl immunostimulant for the treatment of FIP. Polyprenyl Immunostimulant is an investigational veterinary drug. The drug is given orally and it has low toxicity. In the report, three cats with the dry form of FIP were treated with this new drug. Two of the three cats are still on treatment and are alive and well. Once cat was treated for only 4.5 months; that cat survived 14 months, which is pretty long. (He might have survived longer if the owners had continued treatment.)
Once again we have a small, uncontrolled case study. The authors, however, are university professors who are very well known in the veterinary community. Of course, we’ll have to wait for a larger, controlled study before we can make any conclusion regarding whether this is truly is a good treatment option. It’s gratifying to see that there are veterinarians working hard to try to find some type of treatment any type of treatment… for this terrible disease.
(Thanks to catexpert.blogspot.com and Dr. Arnold Plotinick)
Feline Infectious Peritonitis (FIP)
What You Need To know
Dr. Plotinick DVM
Feline Infectious Peritonitis (FIP) is an uncommon, fatal viral disease of cats. It is caused by infection with feline coronavirus (FCoV). There is much confusion and much misinformation about the disease. I hope the information contained in this article addresses most of the concerns and questions that commonly arise, especially when a veterinarian suggests that your cat may have the disease.
Transmission and Development of FIP
Coronaviruses can affect many different species. They cause intestinal and respiratory disease in dogs and pigs, and are a cause of the common cold in humans. Feline coronaviruses are related to these other coronaviruses. The most common form or “Biotype”of FCoV is the enteric (intestinal) form of the virus. This form is easily transmitted between cats. Cats become infected by the fecal-oral route. In other words, FCoV is shed in the feces of one cat, and somehow is ingested by another cat. After the virus is ingested, the virus replicates (i.e. reproduces, making many copies of itself) within the cells that line the intestinal tract. When cats become infected with this form of the virus, they usually show no overt clinical signs. Occasionally, infected cats will get transient, self-limiting diarrhea, probably as a result of the damage to the intestinal cells during viral replication.
Most cats, when they become infected, will shed the virus in their feces for a period of time, usually a few months, and then stop. Infected cats produce antibodies to the virus in response to the infection. Cats may become re-infected with FCoV in the future, and begin shedding again, if they encounter more virus. Again, most encounters are via the fecal-oral route.
Although most infected cats shed the virus for a few months and then stop, a few become persistent shedders, releasing the virus in their feces continuously.
Coronaviruses have an important characteristic: they are fairly prone to genetic change, and seemingly harmless coronaviruses can mutate into a variant virus that may be more pathogenic than the original virus. This, in fact, is how clinical cases of FIP are thought to arise. The relatively benign intestinal coronavirus mutates into a new form type of coronavirus, one that can leave the intestinal tract and cause problems in many different organ systems. This mutated coronavirus is now the deadly, evil FIP virus.
The FIP virus triggers an immune response by the body. The immune response, however, is ineffective and actually makes things worse. Antibodies produced against the FIP virus do not neutralize the virus. The antibodies bind the virus, and these antibody-virus complexes circulate in the bloodstream eventually settling down and accumulating in the walls of the blood vessels where they trigger an intense and harmful inflammatory reaction. These inflammatory lesions can occur in virtually any body system, accounting for the wide range of clinical signs seen in FIP. Normally, when an antibody binds a virus, an immune system cell called a macrophage will gobble up the antibody-bound virus and destroy it. Macrophages, however, don’t destroy the FIP virus, however. Instead, they gobble up the virus and then transport it to other parts of the body, disseminating the disease throughout the body.
Which cats are affected?
Most cases of FIP occur in young cats. In fact, many cases probably occur right after their first infection. This likely has to do with the fact that coronaviruses are prone to mutation. Mutations are more likely to occur when viruses are reproducing themselves, and the highest amount of viral replication occurs right after infection, increasing the risk of the benign intestinal coronavirus transforming into the evil FIP coronavirus. Other stressful factors are often present in young cats, such as an immature immune system, recent re-homing, neutering, vaccination, and perhaps concurrent illness. The peak age for development of FIP is between 6 months and 2 years. Although youngsters fall victim most often, cases are also seen in older cats.
Infection with FCoV is common in cat populations everywhere. Approximately 25 to 40% of household cats are infected with FCoV. In breeding catteries and other large colonies, the number rises to 80 – 100%. Despite this high level of exposure to the virus, FIP is a relatively uncommon disorder. This is because most infections, as stated above, are with the fairly harmless strain of FCoV – the one that produces only mild diarrhea. Cats from breeding catteries and other large multicat households show a higher prevalence of FIP, and this is a reflection of the fact that exposure to FCoV is hard to avoid. In a multicat environment, there may be a few cats who are carriers that continually shed the virus in their feces, reinfecting other cats in the household who also shed the virus, even if only intermittently. In fact, studies have suggested that as few as six cats sharing the same environment are enough to maintain persistent FCoV infections. So, in a multicat environment, the more cats that are chronically shedding FCoV, and the more cats that become reinfected, the higher the risk of FIP developing. Other factors are probably involved, such as genetic susceptibility. A study of all cats diagnosed with FIP over a 16 year period at North Carolina State University revealed that purebred cats were significantly more likely to be diagnosed with FIP, and that Abyssinians, Bengals, Birmans, Himalayans, Ragdolls, and Rexes have a significantly higher risk, whereas Burmese, Exotic Shorthairs, Manxes, Persians, Russian Blues, do not appear to be at increased risk. In colony situations, the incidence of FIP is typically 5%, although this number can vary widely between different colonies. It is not uncommon to see FIP develop in several or al of the kittens from an individual litter over a period of time. In households containing one or two mature cats, the incidence of FIP is very low, perhaps only 1 in 5000.
What are the signs?
Clinical signs associated with FCoV infection are mild – a little diarrhea, perhaps a little vomiting, for a few days duration. FIP, however, can present with a wide variety of clinical signs. There are no clinical signs associated with FIP that are unique for this disease. In general, the disease presents itself in one of two major forms: the effusive (“wet” form, and the non-effusive (“Dry”) form.
As stated above, when the immune system produces antibodies against the FIP virus, the antibodies, rather than neutralize the virus, will bind the virus, and then these antibody-virus complexes circulate in the bloodstream eventually settling down and accumulating in the walls of the blood vessels where they trigger an intense and harmful inflammatory reaction. The inflammation of the blood vessels causes protein-rich fluid to leak through the walls of the blood vessels and accumulate in body cavities. This accumulated fluid is called an effusion, and is the hallmark of effusive or “wet” FIP. On the other hand, the FIP virus can induce an inflammatory response that leads to the formation of clusters of inflammatory cells (called granulomas) accumulating in a variety of organs and structures. The most common sites for granulomas to develop are the abdominal organs such as the liver, kidneys, intestines, and lymph nodes, as well as the eye and the nervous system. The clinical signs that develop depend on which organ or body system is affected. Effusions are not seen in this form of FIP, hence the name non-effusive, or “dry” FIP. “Wet” FIP is more common than “Dry” FIP, and probably accounts for 60 – 70% of FIP cases.
Early in the course of the disease, the clinical signs of both wet and dry FIP tend to be non-specific and include fever, lethargy, poor appetite, and weight loss. The vague nature of these signs is what makes FIP so difficult to diagnose early in the course of disease. As the disease progresses, other clinical signs may become more apparent, perhaps allowing the disease to be categorized as the effusive (wet) or non-effusive (dry) form.
Accumulation of fluid in the abdomen, often causing dramatic abdominal distension, is an obvious manifestation of wet FIP, and is the most common place that fluid tends to accumulate. Less commonly, the fluid will be present in the chest, causing breathing difficulty. Now and then, fluid will be present in both cavities. Occasionally, fluid will accumulate in the pericardial sac (the membrane that surrounds the heart), causing cardiac dysfunction. In many cats with the wet form of the disease, the eyes and the central nervous system will be affected. The liver is commonly affected, sometimes causing jaundice.
Clinical signs of the dry form of the disease can be very vague, making diagnosis a real challenge. Persistent fever that doesn’t respond to antibiotics, progressive weight loss, and lethargy tend to be the main signs. As the disease progresses and granulomas develop, other clinical signs will develop depending on the organ that is affected and the degree of involvement. The FIP virus has a predilection for the eyes and central nervous system and signs of neurologic dysfunction (uncoordinated walking) or eye inflammation are not uncommon.
How is it diagnosed?
FIP is one of the most difficult feline disorders to diagnose. The vague clinical signs early in the course of the disease are seen in many other feline illnesses as well. There are no routine laboratory tests that can definitively confirm one’s suspicions of an FIP diagnosis. Instead, one must evaluate the historical results, clinical signs, physical exam findings and laboratory tests. At this point, one may conclude that they have enough evidence for a presumptive diagnosis of FIP. In cases where the diagnosis remains in doubt, tissue biopsies may ultimately be necessary to make a certain diagnosis.
Historically, cases of FIP are more common in young cats (less than 2 years), purebred cats, and cats from multicat environments.
Routine laboratory tests won’t prove or disprove the diagnosis, but may offer evidence to support or refute one’s suspicions of FIP as the cause of the clinical signs. A complete blood count often shows a low lymphocyte count and high neutrophil count. These are very non-specific changes and are seen in many other illnesses.
A serum biochemistry panel also tends to show non-specific changes, however, elevated globulins are a common and important finding, seen in 50 to 80% of FIP cases. . Hyperproteinemia is usually present, the elevation being due to increase in globulins. Albumin levels may be normal, although frequently they are low or low-normal. Because the liver is an organ that is frequently affected in cases of FIP, elevated liver enzymes and high bilirubin levels are not uncommon.
Coronavirus serology – measuring the level of antibodies against coronavirus in the bloodstream – is common and very misunderstood test. A positive test result simply confirms that the cat has been exposed to a strain of FCoV. Excessive reliance should not be placed on this test. It holds no more value than results of routine hematology and serum biochemistry. Keeping I mind that 25 to 40% of the general cat population and 80 – 100% of cats in multicat households are seropositive for coronavirus, a positive result must be interpreted cautiously. As a general rule, cats with FIP tend to have higher FCoV antibody titers, but there is much overlap in titers between healthy cats that were simply exposed to FCoV vs. sick cats currently suffering from FIP. Again, excessive reliance should not be placed on this test. In fact, there have been cases of FIP in which the coronavirus antibody test showed a lack of detectable antibodies. To summarize: low or medium levels of coronavirus antibodies have no diagnostic value because the virus is so ubiquitous. Negative levels suggest a cat is unlikely to have FIP (although about 10% of cats with FIP will have no detectable antibody); and very high levels of antibody offers support for a diagnosis of FIP, but a strongly positive test is not, in and of itself, diagnostic for the disease.
A valuable diagnostic test is the evaluation of effusions in cases of wet FIP. These effusions tend to be “straw-colored” in appearance and is thick, with a sticky feel to it. The protein content is high, with globulins making up more than 50% of the proteins. There are few diseases that produce effusions with such characteristics. Taken with other supportive evidence, an effusion of this type can provide strong evidence for a presumptive diagnosis, especially if this type of effusion is found in more than one body cavity. Detection of coronavirus particles within the cells that are found in the FIP-induced effusions offers very strong support for a diagnosis (in one study, a positive test was 100% predictive for FIP), but this type of test tends to be performed in research laboratories and most commercial laboratories do not offer this test.
Polymerase chain reaction (PCR) has become a popular high-tech method for detecting the presence of infectious organisms. It does this by detecting minute quantities of viral or bacterial DNA and amplifying it to millions or billions of copies so that it can be detected by a routine laboratory method. Like every other test in veterinary medicine, PCR has its pitfalls, and when it comes to FIP, the PCR suffers from the same limitations as antibody testing – it is unable to distinguish which strain of FCoV to which the cat has been exposed. Those PCRs which claim to have detected the mutation which differentiates the harmless intestinal FCoV from the virulent FIP coronavirus have never been validated by the scientific community.
If significant doubts remain concerning the diagnosis of FIP, a truly definitive diagnosis can be achieved through biopsy of appropriate organs. Biopsy specimens may be obtained via exploratory surgery, or by percutaneous needle biopsy, using ultrasound guidance if necessary. When examined under a microscope by a pathologist, affected tissue samples demonstrate pyogranulomatous inflammation, a characteristic type of inflammation seen in cases of FIP. If there is still doubt as to whether FIP is the cause of the inflammation, additional confirmation can be pursued through immunohistochemistry – using special tissue staining procedures – to detect the presence of FCoV within the biopsy specimen.
Is there any treatment?
FIP is progressive and fatal. Cats with FIP tend to succumb to the disease rather quickly, in a few days or weeks. Wet FIP tends to progress faster than dry FIP. Treatment is generally symptomatic and supportive. Nutritional support, antibiotics, and corticosteroids may produce a temporary alleviation of clinical signs, but the disease invariably progresses. In 2004, a report was published that described the use of recombinant feline interferon and glucocorticoids for the treatment of FIP. Twelve cats with FIP underwent therapy. Four cats succumbed quickly; four cats showed a partial remission (surviving for two to five months), and four cats showed a complete remission (survival time greater than 2 years). The four cats that showed long term survival were older cats – between the age of 6 and 16 – and all had the wet form of the disease. Although the number of cats in the study was small, the results were encouraging in that they suggest an apparent therapeutic effect of recombinant feline interferon in select cases of FIP. However, the high cost associated with feline interferon, and the fact that it is only available in Japan, the UK, and the EC countries may be an obstacle for treatment. Ultimately, nearly all cats diagnosed with FIP are euthanized to alleviate suffering. In terms of contagion, the fact that there have been reports of FIP outbreaks in individual colonies of cats suggests that direct transmission of the FIP-causing coronavirus is occasionally possible, however, the FIP virus does not appear to be commonly transmitted in nature and usually dies out when the affected cat dies from the disease.
Prevention and Control
Because the virus causing FIP is an uncommon mutation of a widespread virus, it can be difficult to prevent and control. The most effective management strategies center around preventing and controlling the prevalence of the underlying infection with FCoV. To prevent FIP in household pets, one should avoid obtaining cats from a source where many cats are housed, and try to obtain them from a place where cats or kittens are kept in small stable groups. To put it more simply, one should obtain healthy kittens/cats from a healthy background. One should avoid having large numbers of cats sharing one living space. Household cats kept singly or in small groups are unlikely to develop FIP.
In breeding catteries, one can attempt to control FIP by reducing the incidence of FCoV infection, or one can try to completely eradicate FCoV within the colony. Eradication of infection, however, is very time consuming and laborious. Maintaining a FCoV-free colony can be quite a challenge, given the ubiquitous nature of FCoV infections. A more realistic approach is to consider eliminating FCoV infection in kittens born into the environment where FCoV is present. If a pregnant queen is isolated from other cats one to two weeks before giving birth, and is then kept isolated with her kittens while practicing good hygiene procedures to prevent environmental spread of infection to the kittens), most of these kittens will remain free of infection. Testing kittens for FCoV antibodies before they are weaned will yield invalid results because kittens derive their antibodies from their mothers, and a positive result merely confirms that the mother was exposed to FCoV. After weaning, however, the queen can be removed and the kittens still kept isolated and tested at 12 – 16 weeks of age for antibodies to FCoV. If the litter tests negative, the isolation procedure has been successful. These kittens are FCoV negative and would theoretically be incapable of developing FIP since they do not have the coronavirus in their body. The isolation procedure sometimes fails if the queen herself is shedding FCoV in her stool and passes it to the kittens. This might be avoided if the kittens are weaned early (at 5 to 6 weeks of age), because the antibodies against FCoV that the kittens obtain from the mother may prevent the kittens of acquiring the infection. Raising FCoV-free kittens requires considerable commitment from breeders.
Good hygiene is always important when trying to minimize the prevalence of FIP. Bearing in mind that the fecal-oral route is thought to be the major way in which the virus is spread, some practical control measures that have been suggested include having enough litter boxes (ideally, one for every two cats), keeping litter boxes away from places where food and water are kept, and keeping all litter boxes very clean. Feces should be removed at least once daily, and litter should be changed as often as practical. Cats should be kept in small stable groups. Regular brushing of the hair coat (especially longhaired cats) is recommended to remove any feces or litter that might get stuck in the fur.
A commercial FIP vaccine is available, however the use of the vaccine is controversial. The vaccine is administered intranasally (i.e.drops in the nose). Different studies of the vaccine have yielded very different results, and the efficacy of the vaccine remains questionable. The American Association of Feline Practitioners and the Academy of Feline Medicine, in their regularly published guidelines for feline vaccination, currently do not endorse the routine use of the FIP vaccine at this time.
Managing the E coli UTI
A urinary tract infection (UTI) occurs when a bacterium, such as uropathogenic Escherichia coli, overcomes host defenses and adheres to, multiplies, and persists in the urinary tract.
Simple UTI
► These infections involve the bladder and are typically initial infections with no concurrent disease or underlying structural or functional abnormality (ie, no compromise of host defense mechanisms).
Complicated UTI
► These infections are associated with pyelonephritis, prostatitis, concurrent systemic diseases (diabetes mellitus, hyper-adrenocorticism), and either systemic (corticosteroid use) or local (indwelling urinary catheter, uroliths, neoplasia) alterations in immunity through compromised host defense mechanisms.
Incidence/Prevalence
E coli is the most common bacterium isolated in canine and feline UTIs.1,2
Approximately 14% of dogs have UTI during their lifetime, but young, healthy cats have an innate resistance and rarely develop UTI.3
Signalment
Age
► Dogs may have UTI at any age.
► Cats older than 10 years of age are predisposed and usually have concurrent illness.
Sex
► Female dogs are at increased risk for UTI due to easier ascension of bacteria through a shorter urethra. In male dogs, UTI is generally classified as complicated.
Causes
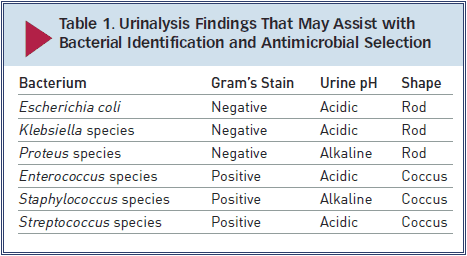
E coli, a gram-negative bacterium, has been isolated in 44% of all canine UTIs and in as many as 67% of feline UTIs.1,4 Other bacteria associated with UTI include Staphylococcus, Streptococcus, Enterococcus,Proteus, and Klebsiella species.
Risk Factors
UTI is associated with alterations in the host’s systemic or local immunity, such as immunosuppression, diabetes, hyperadrenocorticism, anatomic abnormalities (polyps/tumors, recessed vulva), indwelling catheters, uroliths, or urethral sphincter mechanism incompetence.
In cats, risk factors include diabetes mellitus, hyperthyroidism, and renal disease.2
Pathophysiology
E coli is considered a normal component of gastrointestinal and distal urogenital flora, but it can ascend the urethra and gain entrance to the urinary tract. Hematogenous spread is uncommon.
Specific virulence factors found in E coli allow it to adhere to and invade host cells, produce toxins, utilize host nutrients, and evade the host’s immune system.
Signs
History
► Thorough medical history, including details of previous UTI, current or recent medications, and response to treatment, is essential.
► Patient history for UTI typically includes changes in urination (amount, frequency, color, straining). Pollakiuria, stranguria, hematuria, and malodorous urine may indicate cystitis. Dogs with pyelonephritis or prostatitis may have polyuria/polydipsia and signs of systemic illness, such as lethargy, anorexia, and vomiting.
► Some dogs have silent UTIs with no urinary signs, although they may have evidence or history of concurrent disease.
Physical Examination
►Findings on physical examination may be normal in simple UTI.
►Thorough examination for complicated UTI should include abdominal palpation for hepatomegaly, renal pain, thickened bladder wall, and cystouroliths; rectal palpation for prostatomegaly, urethral uroliths, and thickened urethra; and external examination for recessed vulva (Figure 1).
Figure 1. A 1-year-old golden retriever with recessed vulva and evidence of skinfold pyoderma is prepared for episioplasty. Photo courtesy of Emily Klocke, DVM, Diplomate ACVS
DIAGNOSIS
Definitive
Urine culture and susceptibility testing, preferably on a sample obtained by cystocentesis or urinary catheter, are definitive for UTI. Other methods of collection require a quantitative culture to rule out contamination.
Differential
While infection with E coli is most common, many bacteria, including Staphylococcus, Streptococcus,Enterococcus, Proteus, and Klebsiella species, may be responsible. Fungal organisms can also cause UTI.
Laboratory Findings
Complete blood count will be within the reference intervals and is not required for diagnosis of simple UTI. However, an increase in white blood cells may be present with pyelonephritis or prostatitis.
Serum biochemical profile will show abnormalities consistent with the underlying disease (chronic kidney disease, hyperadrenocorticism) in complicated UTI.
Pyuria, bacteriuria (Figure 2), hematuria, or proteinuria may be noted on urinalysis.
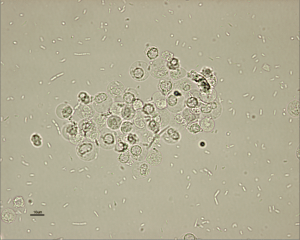
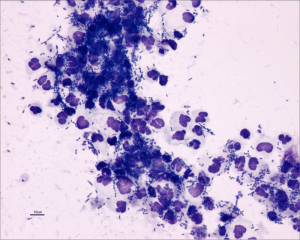
Courtesy of Don Peterson, DVM
► Gram’s staining and measurement of urine pH may help narrow the differential diagnosis while culture is pending (Table 1). However, urine pH may be affected by other factors (fasting versus postprandial, diet, renal disease, vomiting, drugs).
► Not all cases of UTI have active urine sediment; immunosuppression can block the local inflammatory response.
Minimum inhibitory concentrations (MICs) should be requested for culture and susceptibility testing of urine.
Evaluation of endocrine function is indicated if suggested by clinical presentation (measurement of serum thyroxine concentration in a cat with polyphagia, weight loss, and palpable thyroid nodule).
Imaging
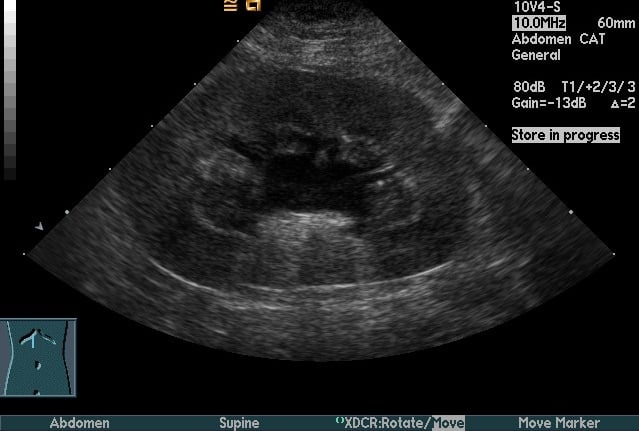
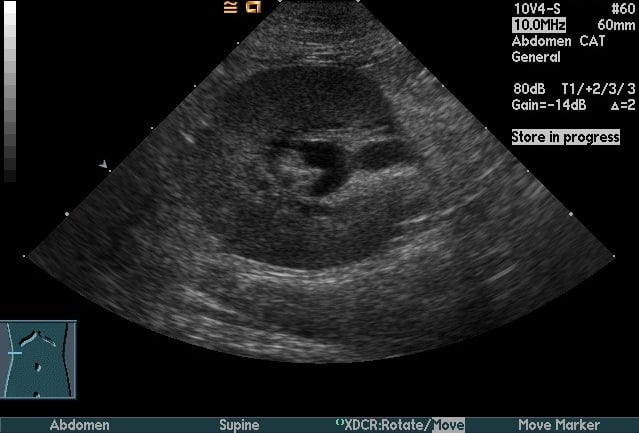
For complicated UTI, radiography and ultrasonography are indicated for assessment of uroliths, pyelonephritis (Figure 3), prostatitis, adrenomegaly, or other complicating factors. Contrast studies or cystoscopy may be indicated to investigate anatomic defects.
Figure 3. Longitudinal (Top) and transverse (Bottom) renal ultrasound images of an 8-year-old female domestic shorthaired cat with pyelonephritis. These images show dilation of the renal pelvis, which is characteristic of pyelonephritis.
TREATMENT
Simple versus complicated infections, location of infection, susceptibility of strain, adverse effects of antimicrobial agents, and client compliance issues should be evaluated prior to treatment for E coliinfections.
Most patients are treated as outpatients, but patients with pyelonephritis or prostatitis due to infection with E coli may benefit from hospitalization and supportive care.
► Most infections can be effectively managed or cured with appropriate antimicrobial treatment, but episioplasty may be indicated in female dogs with a recessed vulva. Surgery or lithotripsy for uroliths or surgical resection of polyps/tumors should be performed when possible.
Concurrent conditions should be identified and managed appropriately to minimize risk for UTI recurrence.
Antimicrobials
Complete 10- to 14-day course for simple UTI or 4 to 6 weeks for complicated UTI.
► Empirical antimicrobial selection can be based on urinalysis findings for simple UTI. Most E coli strains are susceptible to amoxicillin, amoxicillin-clavulanic acid, or cephalexin. Enrofloxacin should be avoided as first-line treatment because of resistance.5
► Antimicrobial choice for complicated UTI should be based on culture and susceptibility testing and the ability of the drug to achieve more than 4 times the reported MIC in urine.
► A lipid-soluble antimicrobial agent that can cross the blood-prostate barrier, such as enrofloxacin, chloramphenicol, or trimethoprim-sulfamethoxazole, is recommended for prostatitis. Castration should be performed after acute inflammation resolves.
Supportive Treatment
Some supportive treatments prescribed in conjunction with appropriate antimicrobial treatment and monitoring (Table 2) may be suitable for patients with recurrent E coli UTI.
Note: Further research is required to determine efficacy, safety, and optimal dosing profiles for these treatments in companion animals.
► Methenamine is a urinary antiseptic that is converted to formaldehyde and can have bactericidal activity against E coli.6 Methenamine is often administered with a urinary acidifier; however it is contraindicated in dogs with metabolic acidosis (chronic kidney disease, diabetic ketoacidosis) and is not well tolerated by cats.
► Proanthocyanidins (found in cranberries) can prevent E coli adhesion to the uroepithelium.7 Preliminary research suggests cranberry extract has antiadhesive effects in canine urine.8,9
► D-mannose is a sugar that competitively binds to mannose-sensitive fimbriae on E coli, which can inhibit adhesion to or displace adhered E coli from the uroepi-thelium.10
► Forskolin is the main active ingredient in the Asiatic herb Coleus forskohlii and may stimulate exocytosis of E coli from the urinary bladder wall.11 It may be useful in relapsed UTI with suspected poor antimicrobial penetration.
FOLLOW-UP
Patient Monitoring
Optimal monitoring for complicated and recurrent UTI includes urine culture 3 to 5 days after initiation of treatment.
► No growth on culture suggests effective treatment.
►Growth on culture suggests ineffective treatment, and antimicrobial choice should be reconsidered.
Bacterial culture should be negative prior to discontinuation of treatment and again 5 to 7 days following discontinuation of antimicrobial treatment.
Recurrence and Resistance
Recurrence of UTI with the same organism warrants further investigation of potential problems with antimicrobial treatment and/or reevaluation for a source of the infection (uroliths or neoplasia).
Recurrence of UTI with a different organism warrants further investigation for a cause of immunosuppression.
Preventive low-dose antimicrobial treatment (eg, nitrofurantoin 4 mg/kg PO once at bedtime instead of Q 8 H) may be indicated for recurrent E coli UTI with an identifiable underlying cause. Low-dose treatment should be initiated after the current infection is properly controlled (following 4�6 weeks of antimicrobial treatment at the therapeutic dose and a negative urine culture).
IN GENERAL
Relative Cost
Simple UTI
►The cost for a physical exam, urinalysis, +/- urine culture & susceptibility, and cost of antimicrobial treatment: $-$
Complicated UTI
►The total expense for initial extensive diagnostic testing, antimicrobial treatment, and multiple follow-up cultures can be costly: $-$
UTI = urinary tract infection
From Clinician’s Brief
Coccida and other Protozoans
Protozoa cause several common feline diseases. Protozoan infections can generally be diagnosed with a fecal float exam by a vet or trained technician. Some of the protozoa are harder to detect and may need special training or stains to diagnose. Here is some information on common protozoan infections:
COCCIDIA
CRYPTOSPORIDIOSIS
TRITRICHOMONAS FOETUS – “Trich”
TOXOPLASMOSIS
GIARDIA CAMPYLOBACTER
COCCIDIA
Coccidia are parasites that are often misunderstood in cats. After the first exposure, coccidia parasites are always present in the kitten’s intestines, and they’re just waiting to take advantage of any digestive upset. It is rarely the initial cause of the diarrhea, but once diarrhea starts, the coccidia will overgrow to large numbers to keep the diarrhea going. Coccidia are a major issue for kittens under 8 weeks old and can even kill them.
Kittens are born with a sterile gut, and their mother seeds their gut with good bacteria during cleaning and care. However, Kittens can also get coccidia from their moms, so the goal is to keep the kitten’s exposure to a minimum number. There are a variety of products you can use for both treatment and prevention, but prevention is most important for consistently healthy kittens.
Cocci Guard is an easy way to prevent coccidia – simply add it to your kitten’s food. The mother should also be fed kitten food with Cocci Guard when you bring her to the birthing area 2 weeks before birth. This decreases the number of coccidia the mother can give to the litter.
Amprolium (Corid or Cocci-Rid) can also be used as coccidia prevention. The flavor is terrible, so some people add it to fish flavored food to try and help the taste, but no flavor works well. The preferred way to use Amprolium is to dose the kitten daily – you know the dose is in and they will also drink adequate water.
Marquis is used to both prevent and treat coccidia. It works well and the once-a-week dose is easy to use. Marquis (ponazuril) will can cause dry eye, so use caution in kittens under 6 weeks old. Marquis lasts 7 days, and when it’s given before shipment, it’s very effective in stopping coccidia from overwhelming the kittens as they adjust to their new home.

We are Michelle and Bill Harrison and we have been breeding and selling kittens together for over 40 years. Kittentanz Cattery is in the foothills of the North Georgia mountains and we specialize in traditional “Applehead” Siamese kittens, and traditional Tonkinese kittens.